AbstractPurposeThe study was conducted to measure stratum corneum hydration (SCH) and pH (SCP) in high-risk newborns in the early postnatal period and to explore the features related to patterns of change in those parameters.
MethodsSCH and SCP were measured on the dorsal hand in 99 hospitalized newborns during the first 14 days of life and the results were analyzed using a general linear model.
ResultsThe mean hydration was 42.9% on day 1, which decreased to 34.6% by 2 weeks (F=15.61, p=<.001). An association was observed between SCH and prematurity (F=21.12, p<.001), as well as for their interaction (F=8.11, p<.001). The mean SCP was 6.2±0.3 on day 1, and decreased to 5.7±0.2 (F=95.75, p<.001), with no association with prematurity. After adjusting for birth weight, SCH was higher in newborns with vaginal delivery (F=9.07, p=.023) and who received phototherapy (F=11.81, p=.011). For SCP, only delivery type had a significant influence (F=6.40, p=.044).
INTRODUCTIONBirth is a very dramatic, deeply real, sometimes chaotic, but nonetheless spontaneous experience in terms of the physiological and ecological adaptions that newborns undergo as they transition from intrauterine to extrauterine life. The ecological adaptation of newborns is characterized by 3 transitions; from aqueous to gaseous, from sterile to septic, and from constant to variable temperature. The skin, the largest and most directly exposed body surface of newborns, is observed, touched, and intervened upon by neonatal clinicians as they provide routine assessments and medical care around the clock during the adaptive transition process.
As high-risk newborns go through a difficult postnatal transition due to inherently high-risk experiences such as preterm birth (PTB) or pathologic conditions, skin integrity plays a critical role as a physical and constitutional barrier against environmental exposure [1]. In newborns, epidermal maturity and the ability to constitute a functional stratum corneum (SC) is inversely proportional to gestational age [1]. Functional integumentary maturity occurs at 33 weeks of gestation [2]. Lower gestational age and birth weight are associated with a greater risk of excessive fluid loss, particularly from the transepidermal surface of the SC, despite the high proportion of fluid by body weight. Newborns confront the challenge of balancing between becoming dry and maintaining hydration of the SC as they emerge from the amniotic fluid during birth. Since physiological maturity of the SC is achieved during the third trimester of gestation [3], preterm newborns are prone to improper levels of SC hydration (SCH). High-risk newborns often are at an increased risk of fluid loss, in addition to their increased demand and dependency on fluid balance due to pathologic conditions such as hyperbilirubinemia, hypoglycemia, and respiratory distress with rapid breathing rate [3]. Hence, SCH may be a critical indicator for assessing fluid balance and even compliance to therapeutic regimens in terms of ecological adaptations to extrauterine life.
The pH of the skin surface is also important for maintaining healthy homeostatic conditions of micro-organisms by inhibiting pathogen overgrowth, as well as for preserving epidermal permeability by activating lipid hydrolases to process lipids and free fatty acids [4]. The functionally appropriate acidification of the SC, known as the acid mantle (AM), occurs during the postnatal ecological adaptive process. The exact mechanism and timing of AM formation on the SC has not been definitively established, but it is known that the pH of the SC (SCP) decreases immediately after birth, after which the AM is formed at a pH range between 4.5 or 5 to 5.5 during the early postnatal period as a natural defensive barrier to promote skin integrity [5]. Although proper acidification of the SC is known to be very valuable for infection control [6], AM formation may be delayed or impaired in premature infants, or in those with inflammatory changes or unusually high hydration of the SC [7].
The skin is the outermost, the largest, the most observed, and the most frequently manipulated body part of hospitalized newborns. High-risk infants are faced with increased risks of impaired or immature skin function, corresponding to nursing diagnoses such as risk for fluid imbalance (diagnostic code 00025), risk for deficient fluid volume (00027, 00028), risk for infection (0004), and risk for surgical site infection (00266) [8]. These nursing diagnoses are identified based on skin conditions as their defining characteristics, and the related factors include age extremes as an at-risk population. As well, studies have reported that skin conditions such as hydration and pH could vary according to endogenous (i.e. sex, age) and exogenous factors (i.e. environmental humidity and exposure) and their interactions in an ecological sense [1,3,4]. Therefore, it is critical for neonatal nurses to understand how parameters such as SCH or SCP vary during the early postnatal period and what factors may influence them.
Fundamental knowledge and understanding of SCH and SCP are vital for assessing the nature and severity of skin-related health concerns, developing a skin care protocol, and evaluating the outcomes of the nursing process to improve skin integrity. However, there is a profound paucity of knowledge about skin parameters and evidence-based clinical protocols for skin care in high-risk newborns as they are suddenly exposed to the gaseous and septic extra-uterine ecology, as many neonatal nurses experience feelings of insecurity regarding neonatal skin care and are uncertain about proper management [9]. The study was conducted to measure SCH and SCP during the early postnatal period in high-risk newborns and to explore features related to their changes during the first 2 weeks of life.
METHODS1. Study DesignA longitudinal, prospective study was designed to measure SCH and SCP in the dorsal hand and to explore features related to their changes in the first 2 weeks of life in high-risk newborns.
2. Setting and SamplesStudy subjects were recruited from hospitalized newborns at a level III neonatal intensive care unit (NICU) in a university-affiliated hospital in South Korea. Newborns transferred from other hospitals with infectious conditions, such as a history of premature rupture of membranes (24 hours or more), sepsis, or skin diseases were excluded from the study, as well as infants with an uncertain birth history or unusual skin conditions.
3. Ethical Considerations and Data CollectionBefore data collection, we obtained Institutional Review Board approval (No: 151208-1A) for conducting the study and provided in-service education on the study to the NICU staff. Once potential subjects were identified in the delivery room, their parents were approached to explain the study to them and to obtain informed consent if they agreed to participate in the study. Subjects were consecutively enrolled from December 2015 to January 2017 in a consecutive manner. Data on SCH and SCP were collected at the dorsal hand on either side 7 times (on days 1, 2, 3, 5, 7, 10, and 14) during the first 2 weeks of life.
SCH was measured using a skin hydrometer (Skin analyzer SK-8, Vcare, Shenzhen, China) at the dorsal hand, with a range of 0.0~99.0% (+1.0%), and SCP was measured using a skin pH meter (HI 99181, HANNA, Woonsocket, RI, USA) in a range of -2 to 16 to 2 decimal points. Environmental humidity and temperature were measured with a hydrometer (TESTO 608-H2, Testo, Lenzkirch, Germany) at the time of data collection at the place where each subject was placed in the NICU, such as the incubator, radiant warmer, or bassinette. The measurements of SCH and SCP were made noninvasively by quickly touching the SC surface with the flat/round end of the tools, and this technique is known not to be risky or harmful. There were no adverse events related to the study during data collection. Demographic (i.e. gestational age and weight) and clinical information (i.e. body temperature, phototherapy, and incubator use) were collected since they are associated with skin conditions and body fluid metabolism. The reliability and validity of the data collection process were evaluated and ensured using a structured manual for procedures, consistent data collection systems, and in-service education for research personnel.
4. Data AnalysisData analysis was performed using IBM SPSS Statistics. During the study period, 99 infants in the NICU met the inclusion criteria of the study and were analyzed, which was a sufficiently large sample size to identify group differences with ⍺=.05 and power of 0.8. Variations in SCH and SCP were analyzed during the 2-week postnatal period using a general linear model that included the clinical characteristics of subjects, such as sex and degree of PTB according to the World Health Organization guidelines, as follows [10]: group 1, severe preterm (gestation<28 weeks); group 2, moderate preterm (28 weeks≤gestation<32 weeks); group 3, mild preterm (32 weeks≤gestation<37 weeks); and group 4, term. In addition to those biological factors, environmental temperature and humidity were included as ecological factors. The relationship between SCP and SCH was analyzed by dividing the participants into 2 groups depending on whether an AM formed (AM-yes group) or not (AM-no group), after which a general linear model (GLM) was applied to analyze SCH variation between those 2 groups.
RESULTSIn the sample of 99 newborns (56 females), the mean gestation was 31.9 weeks (range, 25~41 weeks), the mean birth weight was 1,724 g (range, 590~4,040 g) and the mean Apgar score at 5 minutes was 6.9 (range, 2~10). The distribution of the PTB groups was 8, 36, 47, and 8 newborns in groups 1, 2, 3, and 4, respectively. Sixty-two newborns were born by cesarean section, and 91 were prematurely born.
1. Variations in Hydration and pH of the Stratum Corneum in the First 14 Days of LifeThe mean hydration in the overall sample was 42.9% at the first day of life, which significantly decreased to 34.6% at 2 weeks (F=15.61, p<.001). When hydration change was analyzed by gestational group (Table 1 and Figure 1), it was found that SCH decreased in groups 1 (61.2% to 33.7%) and 2 (49.4 to 37.0%), but increased in group 4 from 23.3% to 33.5% and remained in the 30% range in group 3. A significant difference in the pattern of hydration change was observed by gestational age at birth (F=8.11, p<.001), and significant negative relationships were found between SCH and gestational age (r=-.62, p<.001), birth weight (r=-.53, p<.001), and current weight (r=-.54, p<.001). The mean SCP of 6.2 on the first day of life significantly decreased to 5.7 at 2 weeks (F=95.75, p<.001). However, in contrast to hydration change, dramatic pH variation by groups was not observed, as the pH in all 4 groups decreased for several days of life and then remained at 6.0 to 5.5 (F=1.45, p=.101).
2. Variation in SCH by AM Formation in the First 14 Days of LifeWhen the subjects were divided based on whether an AM formed in the postnatal period (Table 2), the number of infants in the AM-yes group was only 1~5 subjects during the first week and 13 (13.1%) by the 14th day of life, indicating that only 35 of the 693 data points (5.1%) revealed AM formation during the first 2 weeks of life in the 99 participants, even though the proportion of infants with an AM increased over time. In the AM-yes group, clinically significant pattern was hardly identifiable regardless of some variation in hydration, while SCH decreased over time in the AM-no group (F=14.83, p<.001). However, no significant difference in SCH was observed between the 2 groups defined by AM formation when clinical conditions, such as gestational group, delivery type, phototherapy, and environment type (open vs. closed) were adjusted as covariates (F=0.22, p=.650).
3. Changes in SCH and SCP by Clinical Features during the First 2 Weeks of LifeClinical factors related to changes in SCH were analyzed using birth weight (BW) as a covariate, since BW is the indicator that most strongly influences clinical conditions. As presented in Table 3 and Figure 2, SCH was higher in newborns who were delivered normally (F=9.07, p=.023) and received phototherapy (F=11.81, p=.011), and possibly in those who were placed in a closed environment (F=2.79, p=.129). For pH, only delivery type exerted a significant influence (F=6.40, p=.044). All categories of infants showed a decline during the first 2 weeks of life, except for an increase in SCP in newborns placed in an open environment.
4. Comparison of Clinical Characteristics according to AM Formation at the 14th Day of LifeSince only a few subjects (a maximum of 13 newborns) formed an AM during the early postnatal period and once it is formed, it is assumed to be sustainable, the clinical factors associated with AM formation by the 14th day of life were explored (Table 4). The newborns in the AM-yes group were heavier at the time of birth (t=4.61, p=.034) and on day 14 (t=4.76, p=.032) than those in the AM-no group. However, no associations were observed between AM formation and SCH, environmental temperature and humidity, sex, delivery type, PTB, receiving phototherapy, or an open versus closed environment.
DISCUSSIONThis study identified 2 substantial findings on hydration change and AM formation in high-risk newborns after birth. First, for high-risk newborns, the general standard for SCH may be the 30% range, independently of clinical conditions such as birth experience, receiving phototherapy, or being placed in an open or closed environment. Yosipovitch et al. [11] reported various measurements of skin hydration, from approximately the 30% range to the 80% range, at different body areas for 1~2 days after birth. A study [1] reported that SCH generally was in the 50~80 range on a scale extending from 0 (maximum dryness) to 150 (maximum humidity) on the third day of life in healthy term newborns. Furthermore, Visscher et al. [12] reported that SCH tended to plateau at 10~15 days in both the diapered and non-diapered regions in term newborns. In our previous studies [13], the hydration level in premature newborns ranged from 32.7% to 33.9% at 7 days of life in premature infants, while the hydration level was 36.4% at 15.3 days of life in premature newborns and 34.1% at 10.3 days in full-term newborns. Similarly, in our current study, while SCH ranged from the 20% to 60% range on the first day, it decreased or increased to converge in the 30% range by 2 weeks of life. The starting point of SCH varied by gestational age at birth, and shorter gestations were associated with higher SCH. Moreover, our findings showed that higher SCH values were observed in high-risk newborns who were delivered normally, received phototherapy, and were placed in a closed environment, such as an incubator, than their counterparts. These findings were obtained from an analysis that was adjusted for BW, which might have been too primitive to make a decisive conclusion about the factors influencing SCH. However, regardless of whether SCH increased or decreased during the first 2 weeks of life, it consistently converged toward the 30% range. As observed in this study, the inverse relationship of SCH with gestational duration is well known [1]. This relationship can be explained based on the fact that a shorter gestational period leads to a higher level of transdermal water with a limited capacity for keratinization [3], as well as a higher likelihood of placement in an area with a high level of environmental humidity. However, it is not certain what role gestation plays on the keratinization process of SC or whether higher levels of SCH might simply postpone the keratinizing process physically. However, our study findings confirm that the reference level of SCH is the 30% range, which should be achieved within 2 weeks of life, even in high-risk newborns. That is, the standard reference level of SCH should be the 30% range for newborns, although it may take newborns with risk factors longer to reach that range. It is quite remarkable that SCH converges in the 30% range, whereas the permeability of the SC and the duration of water (amniotic fluid) immersion varies depending on the clinical conditions of newborns, such as gestational age or pathologic conditions.
The second finding is that AM formation is very unlikely in the first 2 weeks of life in neonates with high-risk conditions. The acidified layer of the SC plays a defensive role against microbial outgrowth on the body surface [6]. While the functional pH range of AM is known to range from 4.5 to 5.5 [5], the timing and the exact pH values of the AM in newborn subjects are subjective and inconsistent. Some studies have reported less acidic skin in infants (pH, 5.45~6.06) than in adults [14]. A study reported a slightly higher pH in the diaper area (5.88 versus 5.65) and chest area (5.39 versus 5.31) in full-term newborns than in premature newborns in the NICU [7]. Fluhr and Elias [15] summarized variation in skin pH at different ages and sites in human, stating that the pH is around 5 at dry sites, such as the forearm or leg/back, around 6 at wet sites, such as axillary or inguinal regions, and around 6~7 for the first few days of life. A study of healthy preterm newborns (gestational days: 212~257) reported that the SCP was 5.65 on the second day and 5.47 at the second week of life at the upper leg, and 5.31 on the second day and 5.16 at the second week of life at the forehead [16]. Our previous study [13] reported a similar pH value (5.48 on the dorsal hand) and that 57.1% of subjects had formed an AM by the seventh day of life. These findings indicate the possibility of wide pH ranges in the SC according to the measurement site and depending on the clinical conditions of newborns.
In this longitudinal study conducted to observe the daily adaptive patterns of skin pH after birth, only 2 of 99 subjects achieved an AM on the dorsal hand by the second day, and 13 did so by the 14th day. The infants in the AM-yes group had a greater gestational age and BW than those in the AM-no group, which might explain this observation. This possibility is supported by our previous study [13], as well as that of Kanti et al. [16], in which the subjects had healthier BWs and a greater gestational age at birth than the subjects of our study. The pH of the outer skin is determined by adaptive interactions between the body and the environment. A study [15] reported that 3 types of factors influenced SCP in humans; endogenous factors, pathologic conditions, and exogenous factors. The subjects of this study were high-risk newborns hospitalized in the NICU; 44% of our subjects were born before 32 weeks of gestation, including 8 subjects born before 28 weeks of gestation.
The absence of AM formation in most subjects (86 newborns) on the 14th postnatal day suggests that the skin of these newborns had a limited capability for AM formation. Moreover, AM formation was not related to clinical or exogenous features such as SCH, delivery type, phototherapy, or an open versus closed environment. While we reported a positive correlation between pH and SCH in a previous study [17], in the present study, we did not identify a significant difference in SCH depending on AM formation, despite a tendency for AM formation to be associated with SCH in an analysis adjusted for gestational group, delivery type, phototherapy, and environment type as covariates. One possible explanation for not observing a relationship between SCH and AM might be the small number of subjects in the AM-yes group, leading to unequal variance between groups with an impact on statistical robustness. Nevertheless, our study clearly demonstrated that NICU newborns have a limited ability to form an AM, which might be due to intrinsically high-risk factors, such as PTB, or pathologic conditions. As the AM plays an important protective role as a skin barrier, the less acidic surface pH of newborns is related to the incomplete formation of a barrier against infections [6,18]. This study underscores that the early vulnerability of high-risk newborns to infections might be due to their low likelihood of having an AM on the outer skin, in addition to their high-risk characteristics.
CONCLUSIONNewborns experience very dramatic environmental changes immediately after birth, including the transition to a shared, septic, aerial space from the alkaline and sterile amniotic fluid that they are exclusively exposed to in intrauterine life. Infections and the risk thereof are one of the primary nursing concerns for neonates, and maintaining skin integrity is an action goal for hospitalized newborns undergoing the ecological adaptive process to the extrauterine environment. Understanding SCH and AM formation can contribute to the development of evidence-based, solid skin care protocols for nursing professionals. This study found that SCH in the 30% range could be achieved by 2 weeks of life, and that AM formation within 2 weeks of life was extremely unlikely in newborns with high-risk conditions. These findings could serve as a reference for evidence-based nursing practices in applying nursing processes to improve skin integrity in high-risk newborns. Further study is needed to identify how long it takes for the AM to form in these infants, the relevant mechanisms and influencing factors. and the definitive relationship between pH and infection risk, particularly when newborns are faced with various exogenous conditions.
AcknowledgementsWe would like to express special thanks to our research assistants for data collection; Namhee Kim, Kyunghee Son, and Eunmi Jung.
REFERENCES1. Hoeger PH, Enzmann CC. Skin physiology of the neonate and young infant: A prospective study of functional skin parameters during early infancy. Pediatric Dermatology. 2002;19(3):256-262. https://doi.org/10.1046/j.1525-1470.2002.00082.x
2. Baharestani MM. An overview of neonatal and pediatric wound care knowledge and considerations. Ostomy Wound Management. 2007;53(6):34-36.
3. Visscher MO, Adam R, Brink S, Odio M. Newborn infant skin: Physiology, development, and care. Clinics in Dermatology. 2015;33(3):271-280. https://doi.org/10.1016/j.clindermatol.2014.12.003
4. Fluhr JW, Kao J, Jain M, Ahn SK, Feingold KR, Elias PM. Generation of free fatty acids from phospholipids regulates stratum corneum acidification and integrity. Journal of Investigative Dermatology. 2001;117(1):44-51. https://doi.org/10.1046/j.0022-202x.2001.01399.x
5. Dyer JA. Newborn skin care. Seminars in Perinatology. 2013;37(1):3-7. https://doi.org/10.1053/j.semperi.2012.11.008
6. Elias PM. The how, why and clinical importance of stratum corneum acidification. Experimental Dermatology. 2017;26(11):999-1003. https://doi.org/10.1111/exd.13329
7. Visscher MO, Taylor T, Narendran V. Neonatal intensive care practices and the influence on skin condition. Journal of the European Academy of Dermatology and Venereology. 2013;27(4):486-493. https://doi.org/10.1111/j.1468-3083.2012.04470.x
8. Herdman HT, Kamitsuru S. Nursing diagnoses: Definitions and classification 2018-2020. 11th ed. Stuttgart: Thieme; 2017. p. 184-383.
9. Liversedge HL, Bader DL, Schoonhoven L, Worsley PR. Survey of neonatal nurses' practices and beliefs in relation to skin health. Journal of Neonatal Nursing. 2018;24(2):86-93. https://doi.org/10.1016/j.jnn.2017.07.007
10. Euser AM, de Wit CC, Finken MJ, Rijken M, Wit JM. Growth of preterm born children. Hormone Research in Paediatrics. 2008;70(6):319-328. https://doi.org/10.1159/000161862
11. Yosipovitch G, Maayan-Metzger A, Merlob P, Sirota L. Skin barrier properties in different body areas in neonates. Pediatrics. 2000;106:105-108.
12. Visscher MO, Chatterjee R, Munson KA, Pickens WL, Hoath SB. Changes in diapered and nondiapered infant skin over the first month of life. Pediatric Dermatology. 2000;17(1):45-51. https://doi.org/10.1046/j.1525-1470.2000.01711.x
13. Ahn Y, Sohn M, Lee S, Lee M. pH, temperature, hydration of the stratum corneum of the dorsal hand and the cord area, and acid mantle formation during early days of life in high-risk newborns. Journal of Korean Academy of Nursing. 2013;43(1):30-38. https://doi.org/10.4040/jkan.2013.43.1.30
14. Giusti F, Martella A, Bertoni L, Seidenari S. Skin barrier, hydration, and pH of the skin of infants under 2 years of age. Pediatric Dermatology. 2001;18(2):93-96. https://doi.org/10.1046/j.1525-1470.2001.018002093.x
15. Fluhr JW, Elias PM. Stratum corneum pH: Formation and function of the 'acid mantle'. Exogenous Dermatology. 2002;1(4):163-175. https://doi.org/10.1159/000066140
16. Kanti V, Bonzel A, Stroux A, Proquitté H, Bührer C, Blume-Peytavi U, et al. Postnatal maturation of skin barrier function in premature infants. Skin Pharmacology and Physiology. 2014;27(5):234-241. https://doi.org/10.1159/000354923
17. Ahn Y, Sohn M, Jun Y, Lee E, Lee S. Two methods of cord care in high-risk newborns: Their effects on hydration, temperature, pH, and floras of the cord area. Journal of Child Health Care. 2015;19(1):118-129. https://doi.org/10.1177/1367493513503580
18. Behne MJ, Barry NP, Hanson KM, Aronchik I, Clegg RW, Gratton E, et al. Neonatal development of the stratum corneum pH gradient: Localization and mechanisms leading to emergence of optimal barrier function. Journal of Investigative Dermatology. 2003;120(6):998-1006. https://doi.org/10.1038/jid.2003.11
Figure. 1.Comparison of changes during the first 14 days of life in the hydration and pH of the stratum corneum among prematurity groups. (Group 1=For severe preterm (gestation<28 weeks); Group 2=For moderate preterm (28 weeks≤gestation<32 weeks); Group 3=For mild preterm (32 weeks≤gestation<37 weeks); Group 4=Term gestation at birth.) 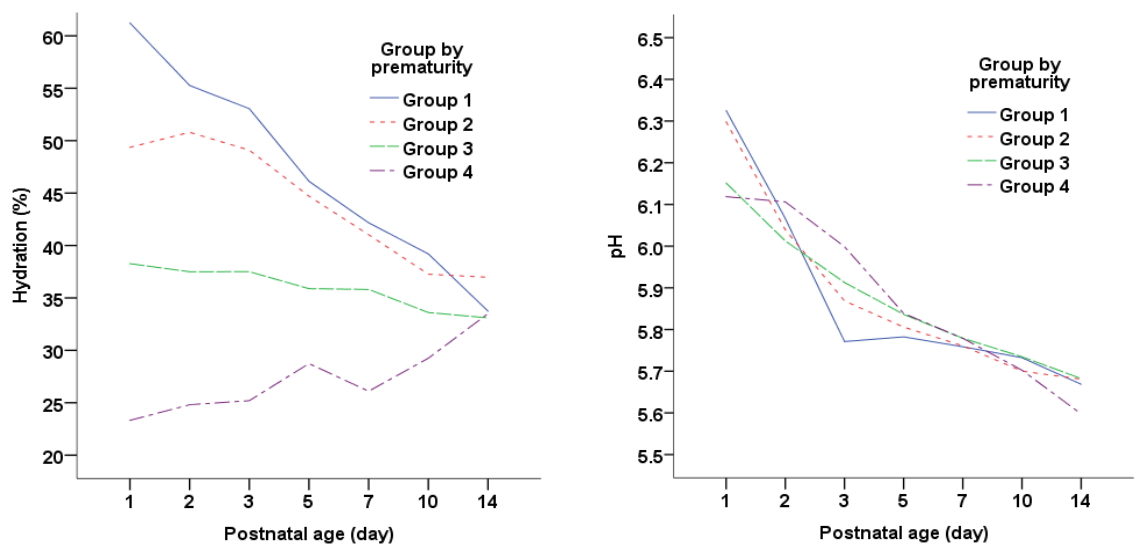
Table 1.Changes in Hydration and pH of the Stratum Corneum of the Dorsal Hand during the First 14 Days of Life (N=99) Table 2.Hydration Changes of the Stratum Corneum by Acid Mantle Formation on the Dorsal Hand during the First 14 Days of Life (N=99)
Table 3.SCH Change by Clinical Features during the First 2 Weeks of Life (N=99)
Table 4.Clinical Comparison between AM Formation Groups at the 14th Day of Life (N=99)
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||